
In this article, you will discover and understand reducing agent for Reverse Osmosis Systems. Reducing agent known as also Reductant for RO membrane. So, water treatment is a critical process in various industries, and reverse osmosis (RO) systems have emerged as a reliable and cost-effective solution. Meanwhile, the success of an RO system depends on several factors, including the proper design and optimization of the system, as well as the selection and effective use of reducing agents. In this comprehensive guide, we will explore the role of reducing agents in RO systems, their chemistry, and their applications.
Understand Oxidizing and Reducing Process
In looking at oxidation-reduction reactions, we can focus on the role played by a particular reactant in a chemical reaction. What is the role of the permanganate ion in the following reaction, for example?
Oxidation: H2C2O4 ——> CO2
The permanganate ion removes electrons from oxalic acid molecules and thereby oxidizes the oxalic acid. Thus, the MnO4- ion acts as an oxidizing agent in this reaction. Oxalic acid, on the other hand, is a reducing agent in this reaction. By giving up electrons, it reduces the MnO4– ion to Mn+2.
I. Introduction to Reverse Osmosis Systems
Reverse osmosis is a water purification method that utilizes semi-permeable membranes to remove impurities and contaminants from water. By applying hydraulic pressure, water molecules are forced through the membranes, leaving behind dissolved salts, organic materials, and other contaminants. So, RO systems offer numerous benefits in industrial applications, such as power generation and water treatment.
II. Understanding Reducing Agents for Reverse Osmosis
Reducing agents play a crucial role in the proper functioning of RO systems. So, these agents are substances that have the ability to donate electrons, thereby reducing the oxidation state of other substances. In the context of RO systems, reducing agents are used to break down chlorine and other oxidative compounds that can be detrimental to the performance of the system.
III. The Chemistry of Reducing Agents
The most commonly used reducing agents for Reverse Osmosis Systems is sodium bisulfite (NaHSO3). When injected into the water stream, sodium bisulfite reacts with free chlorine, converting it into harmless chloride ions, accordingly. So, this reaction involves the donation of electrons from the reducing agent to the chlorine atoms, resulting in the breakdown of chlorine.
Sodium Bisulfite (NaHSO3) is a reducing agent capable of dechlorinating the feed water to a Reverse Osmosis System.
Dechlorination reactions:
NaHSO3 + Cl2 + H2O —-> NaHSO4 + 2HCL
1ppm, of Chlorine (Cl2) reqires 1.46ppm of NaHSO3
To ensure complete Dechlorination add 10% excess of sodium bisulfite
- Sodium Bisulfite (NaHSO3) is a reducing agent capable of dechlorinating the feed water to a Reverse Osmosis System.
- Dechlorination reactions:
NaHSO3 + Cl2 + H2O —-> NaHSO4 + 2HCL - 1ppm, of Chlorine (Cl2) reqires 1.46ppm of NaHSO3
- To ensure complete Dechlorination add 10% excess of sodium bisulfite
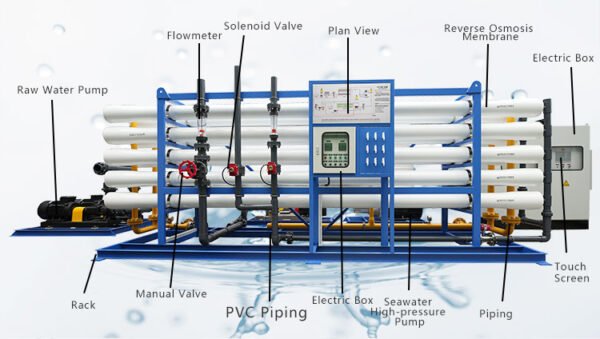
IV. Applications of Reducing Agents in RO Systems
Reducing agents are primarily used in two critical areas of RO systems: pre-treatment and post-treatment. First, tn the pre-treatment stage, reducing agents for reverse osmosis plant are employed to protect the RO membranes from the detrimental effects of chlorine and other oxidative compounds present in the feedwater. Second, in the post-treatment stage, reducing agents are used to eliminate residual chlorine and ensure the purity of the treated water.
A. Pre-Treatment with Reducing Agents
In the pre-treatment stage, the main objective is to protect the RO membranes from incompatible substances, particularly chlorine. Chlorine can have a detrimental impact on the performance and lifespan of RO membranes. By injecting reducing agents such as sodium bisulfite, the chlorine is effectively broken down into harmless chloride ions, preventing membrane damage.
B. Post-Treatment with Reducing Agents
After the RO process, residual chlorine may still be present in the treated water. So, this residual chlorine needs to be eliminated to ensure the water’s purity and prevent any adverse effects on downstream processes or applications. Reducing agents are used in post-treatment to completely remove residual chlorine, providing a final step in the water purification process.
V. Maintenance and Considerations for Reducing Agent Usage
To ensure the effectiveness of reducing agents for Reverse Osmosis systems, proper maintenance and considerations are essential. Here are some key factors to keep in mind:
A. Dosage and Injection
The proper dosage of reducing agents, such as sodium bisulfite, is crucial for effective chlorine breakdown. So, the reducing agent should be injected whenever the RO inlet valve opens, including during filling or flushing operations. So, the injection point should be immediately upstream of the inlet isolation valve to minimize chlorine diffusion during shutdown.
B. Potential Challenges and Solutions
While reducing agents offer effective chlorine breakdown, certain challenges may arise. It is important to address these challenges promptly to prevent any adverse effects on the RO membrane elements. Potential challenges include the depletion of reducing agent solution, inadequate injection pump settings, and insufficient mixing of the reducing agent with the feedwater. Regular monitoring and maintenance can help mitigate these challenges.
C. Carbon Filtration as an Alternative
Activated carbon filtration can serve as an alternative to reducing agent injection for chlorine breakdown. Activated carbon has a high capacity for chlorine removal through an electrochemical reaction. This method offers a reliable means of breaking down chlorine and can be a more cost-effective option.
D. Maintenance Considerations
Regular maintenance is critical to the success of reducing-agent injection or carbon filtration. Activated carbon media should be replaced annually or based on an increase in the effluent concentration of total organic carbon (TOC) to prevent the shedding of biological particles into the RO system. Over-injection of reducing agents can lead to increased breakdown of dissolved oxygen in the water, potentially causing bacterial growth and fouling. It is important to maintain a residual reducing agent concentration within the appropriate range to minimize these risks.
VI. Advantages of Reducing Agents in RO Systems
The use of reducing agents in RO systems offers several advantages, including:
- Protection of RO membranes from chlorine and oxidative compounds
- Prevention of membrane damage and fouling
- Enhanced water purity through the elimination of residual chlorine
- Cost-effectiveness compared to alternative methods of chlorine removal
- Maintenance of optimal system performance and longevity
What are 3 examples of reducing agents?
Examples of substances that are common reducing agents include the alkali metals, formic acid, oxalic acid, and sulfite compounds. In their pre-reaction states, reducers have extra electrons (that is, they are by themselves reduced) and oxidizers lack electrons (that is, they are by themselves oxidized).
How do you determine the best reducing agent?
Strong reducing agents easily lose (or donate) electrons. Atoms with relatively large atomic radii tend to be better reductants. In such species, the distance from the nucleus to the valence electrons is so long that these electrons are not strongly attracted. These elements tend to be strong reducing agents. Good reducing agents tend to consist of atoms with a low electronegativity, the ability of an atom or molecule to attract bonding electrons1, and relatively small ionization energies serve as good reducing agents too. “The measure of a material to oxidize or lose electrons is known as its oxidation potential”.
VII. Conclusion
Reducing agents play a vital role in the successful operation of reverse osmosis systems. By effectively breaking down chlorine and other oxidative compounds, reducing agents protect RO membranes and ensure the purity of treated water. Proper dosage, maintenance, and consideration of alternative methods such as activated carbon filtration are essential for maximizing the benefits of reducing agents in RO systems. By understanding the chemistry and applications of reducing agents, industries can optimize their water treatment processes and achieve efficient and reliable results.
