What is the fouling and scaling on membrane? Proven High Tech Solutions:
Fouling and scaling on membrane filtration are one of the biggest challenges in water treatment industry. Today, I will explain to you how to understand your source water can make fouling or scaling on your membrane filtration. And how you can use longer your membranes?

First of all, we explained what is fouling and fouling types and scaling.
What is the meaning of fouling in water treatment?
You know, reverse osmosis is pressure driven process. So, we make pressure on water and force it to pass semi permeable membrane. The fouling of pressure-driven membranes is generally referred to the accumulation, deposition, or adsorption of foulants onto the surface of membrane or within the membrane pores. Which can cause the basic membrane functions not work properly such as over filtration time, reducing permeate flow, filtration efficiency, and pressure drop across the membrane.
Generally, when permeate flow rate reduces, water plant operator increases the pressure, but it causes increasing the fouling speed at membrane. So, we do not use membranes as long as possible. Do not worry, we will explain step by step and you will learn how to use membrane longer. But first of all, you have to understand mechanism of fouling and scaling.
Membrane fouling is influenced by three major factors:
- membrane material properties (e.g. hydrophilicity, roughness, and electrical charge),
- water character depends on the feed water analysis report (e.g. the nature and concentration of the foulant)
- operating conditions (e.g. high-pressure operation increases fouling).
Here, I want to tell something, we have face this problem so often at customer side. I understand customer wants to get best price in market. And if customer does not have enough knowledge about reverse osmosis system, he just tell to supplier needed capacity, as liter per hour. Most of professional water treatment supplier, first ask what is water source and do you have water analysis report?
If customer does not understand the meaning of how this question important, just focus on price and try to buy cheapest solution, they are buying wrong system. After using the system, maybe fouling, maybe scaling problem occur, flow rate dramatically reduce in short time and operator increase the pressure, membrane was clogged and cannot be used again, maybe high pressure pump is also corrupted,. Then customer is thinking, China Product Quality is not good, of course if they buy from china. As Chunke, we never give proposal, if customer does not have water analysis report.
“good things are not cheap and cheap things are seldom good.”
Fouling Types in Reverse Osmosis Water Filtration:
As you can see in this picture, there are 3 types of fouling in the membrane. Biofouling, colloidal fouling, and organic fouling. Now, we see what are they? Fouling and scaling on membrane filtration is important problem.
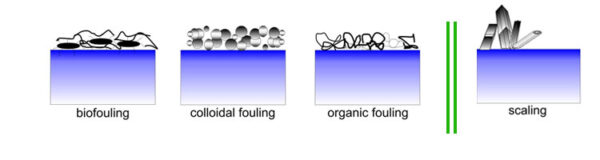
Biofouling on ro membrane:
Biofouling is very closely relating to the living microorganisms in the feed water. So that attach onto the membrane surface. In general, the attached microorganisms tend to grow rapidly by forming biological film at the presence of nutrients in the feed water. So, biofouling put another barrier on membrane and increase the membrane resistance. In the same time, biofilm causes hydrolysis process in membrane layer, it means polymer degradation, kill your membrane.
Colloidal Fouling:
Collodial fouling occurs when suspended solids and – or colloidal material clog the holes of a membrane or stick to its surface. As particles accumulate on the membrane, they form a “cake” layer that obstructs water from flowing through the membrane’s pores, as a result it increases pressure differential measurements and increases energy consumption.
Organic Fouling:
Organic fouling is the combination of deposition, reactions, and interactions of high-molecular-weight organic molecules, for example, natural organic matters with the membrane surface. These organic matters usually consist of humic substances, polysaccharides, proteins, lipids, nucleic acids, and amino acids, organic acids, and cell components.
As you see, fouling is coming from organic matters, scaling is a kind of fouling because of inorganic matters.

What is the meaning of scaling in water treatment?
Scaling is coatings on the membrane, formed by inorganic crystallization. Usually, they only occur with NF and RO membranes if the solubility limit of dissolved salts is more than standard. You can see hard, crystal form coat layer on membrane.
How to minimize Fouling and Scaling on Membrane Filtration?
You understand what is fouling and scaling and now we can talk about how we can minimize if there is organic and inorganic chemicals cause fouling or scaling in your feed water.
First thing , you have to do, you send your water to laboratory for test and get the test result. Then be sure, your water can cause fouling or scaling in your ro plant.
Fouling and scaling can minimize by;
– Appropriate membrane selection,
– Choice of operating conditions,
– Pretreatment system,
– Membrane cleaning system, CIP (clean-in-place)
Appropriate membrane selection:
When check the test report, as regards to your water source and Total Dissolved Solids, TDS. If you do not know TDS, please watch this video. You can decide which membrane type is suitable for your application. If your water is brackish water, you must choose brackish water type membranes. If your water source is seawater, you must choose seawater types membranes.
Choice of operating conditions
When we check your water analysis report, we design system as regards to working pressure and flow rate and you have to follow our design operation conditions. To increase working pressure To get more yield from system can cause fouling and scaling on membrane filtration.
Pretreatment system
Also, when you check your water analysis report, you will see TSS (total suspended solids) and Turbidity color value. Now we see, what is the effect of this value on water.
Turbidity and TSS can cause fouling problem on membrane and applying pretreatment such as coagulation, flocculation, sand filter, carbon filter remove suspended solids and matter in your water and prepare water to membranes.
Membrane cleaning system, CIP (clean-in-place)
The other method is CIP system to clean, wash membrane in membrane housing, without taking them out, automatically. You can use acids and bases to remove foulants.
How to understand your water has scaling problem?
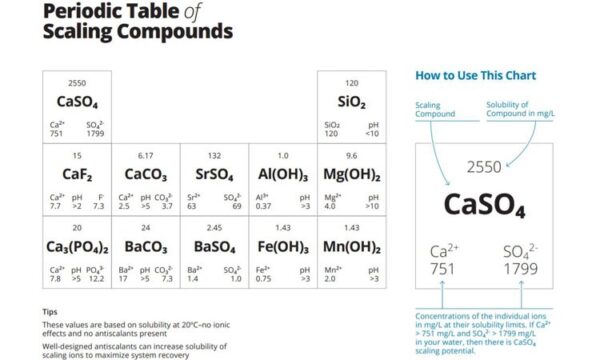
Here there is a table, how we will use this table. Please follow carefully. You have water analysis report. And you want to understand, is there any scaling problem in your membrane. To happen Calcium sulphate scaling, our calcium level must be higher than 751mg/L or ppm, and sulphate must be higher than 1799mg/L or ppm. So from test report, our calcium is 1020ppm, sulphate is 9950ppm, so there is calcium sulphate scaling problem. Or we check others, aluminum hydroxide, in test report, aluminum is 0.5mg/L, it is higher than 0.37ppm, and pH is 7.5, it is higher than 3, so there is potential for aluminum hydroxide scaling. Such as now we check is there any potential for Stronium sulphate scaling, Sulphate is 9950mg/L, it is higher than 69ppm, but strontium is just 22ppm, it is less than 63, so we do not have this risk.
To minimize scaling and fouling, there are a lot of chemical choice in the market, to suggest which one is fix to your problem, we have to see your water analysis report.
Depends on the fouling type, cleaning agents are applied. For example, for scaling and colloidal fouling, acids, e. g. citric acid, acetic acid is applied. For organic fouling, anionic surfactants, oxidants, e. g. hypochlorite, hydrogen peroxide, or alkaline cleaning agents, e. g. caustic soda solution. And for biofouling, disinfectants, e. g. hypochlorite; biocides can be applied.
How to control scaling problem in reverse osmosis water plant?
In fact, scale control is complex but the particular procedure depends on the composition of the feed water.
Calcium carbonate scale, by far the most common problem, is easily controlled by acidifying the feed or by using an ion exchange water softener to exchange calcium for sodium.
Alternatively, an antiscalant chemical such as sodium exametaphosphate can be added. Because antiscalants interfere with the precipitation of the insoluble salt and maintain the salt in solution even when the solubility limit is exceeded.
Polymeric antiscalants may also be used, sometimes in combination with a dispersant to break up any flocs that occur.
Once the silica scale formed, it is difficult to remove because no effective antiscalant or dispersant is available. The solubility of silica is a strong function of pH and temperature, but in general, the brine should not exceed 120 ppm silica. Once formed, the silica scale is difficult to remove.
Acid cleaning agents such as hydrochloric, phosphoric, or citric acids effectively remove common scaling compounds.
With cellulose acetate membranes the pH of the solution should not go below 2.0 or else hydrolysis of the membrane will occur.
Oxalic acid is particularly effective for removing iron deposits.
For more information, please contact with us: info@chunkewatertreatment.com

